In this experiment, HENK fashioned a thick INOX wire to explore the possible magnetic effects on the plasma. The aim was to 'run' the supply current of 1.5 amps through the wire to the anode to create a small magnetic field and see what would happen. The steel ball is 15mm and produces good feedback in the present vessel.
With Argon in the tank a nice argon plasma colour is achieved. Notice that the self organised plasma features face the away from the core of the anode geometry in the direction of the cathode.
Immediately, we can see there are both stable and unstable self-organised plasmas on the INOX anode wire coil. The gif animation below makes this clear.
Moreover, if we look at the self-organised plasma features, we see that there appears to be one or two brighter sub-structures in them. In the below gif animation I have done some contrast adjustment to highlight this.
In this differently framed video, we see that there is some periodic event on the aluminium part of the cathode in the background HENK referred to as ‘the pulsar’. Note this 27Al cathode sheet degraded quite quickly.
The intervals actually grows as time passes which can be seen on the markers below
Number of 1/30s frames between consecutive pulse starts | Frame growth of interval
196
214 | +18
226 | +12
241 | +15
255 | +14
270 | +15
Adding a little Hydrogen into the atmosphere makes the plasma more orange and you can see a nice white surface edge plasma on the lower part of the wire. The temperature of the plasma will lead to the production of atomic hydrogen.
In this video, you can see what may be the kind of anomalous metal luminosity that has been seen in a range of experiments, especially those where Hydrogen or water vapour are in play with high frequencies. Typically, the metal is yellow/orange and can even be incandescent, but it does not necessarily mean the metal is at the corresponding colour temperature.
This was seen by John Hutchison, where a ‘rat tail file’ glowed as if very hot, but it did not even make a mark on the wooden supports it was on and was cool to the touch shortly after it split in two.
LENR researcher ‘me356’ was able to turn this anomalous metal glowing on and off in a few seconds in stainless steel, without changing the temperature of the reactor. The researcher noted that stainless steel is not a good thermal conductor.

me356 was using metals known to catalyse the production of atomic hydrogen.
[For more information, refer to this blog]
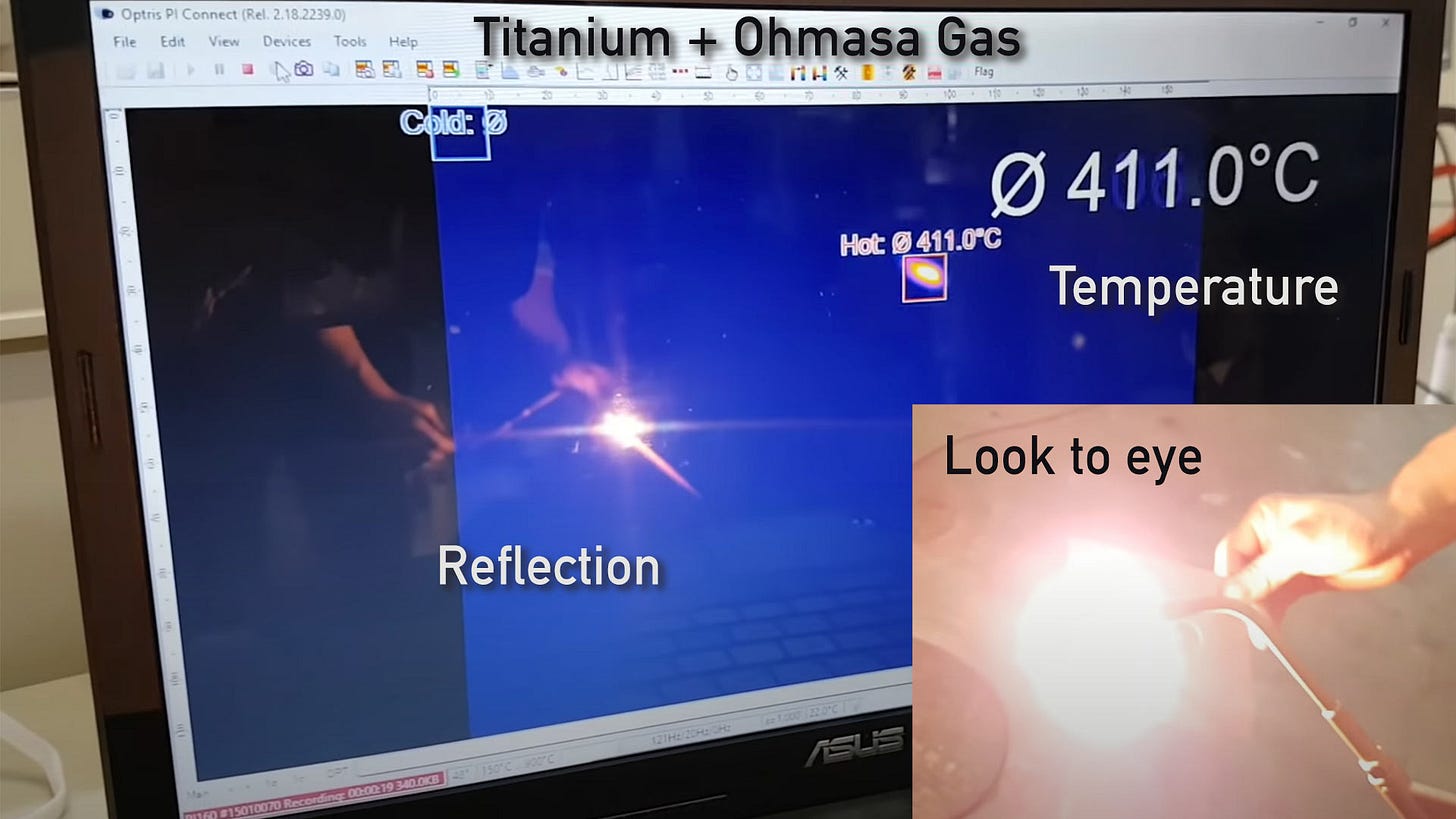
So-called HHO or Brown Gas has been shown to do the same thing. Last year, I had the opportunity to test Ohmasa Gas (which has been shown by analysis to contain atomic hydrogen) on a range of different metals with an Optris PI160 Thermal Imaging camera and was able to show that metals such as Titanium and Tungsten glow incandescently WELL BELOW the temperature at which they should.
So what do you think? Is the 15mm ball almost immediately getting hotter? Or is it a surface or bulk electron collective excitement? Is it something else?






Nice, Merry Xmas Bob :)
So is the reactor as simple as a metal anode within a steel chamber fill with gas at a high electrical potential difference ? Or is there more to it ?